 However, his model worked well as an explanation for the emissions of the hydrogen atom, but was seriously limited when applied to other atoms. Learning Objectives To know the relationship between atomic spectra and the electronic structure of atoms. Moving up the ladder increases your potential energy, while moving down the ladder decreases your energy.īohr's work had a strong influence on our modern understanding of the inner workings of the atom. Im new to thingiverse and Im not sure how or even if its possible to include colors. The solar system or planetary model of the atom was attractive to scientists because it was similar to something with which they were already familiar, namely the solar system. As you move up or down a ladder, you can only occupy specific rungs and cannot be in the spaces in between rungs. An everyday analogy to the Bohr model is the rungs of a ladder. Download this Bohr Model Of Carbon Atom With Proton Neutron And Electron photo now. To make a Bohr model of carbon, you must check the Periodic Table to see how many protons, neutrons, and electrons carbon has. The electron is not allowed to occupy any of the spaces in between the orbits. The orbits that are further from the nucleus are all of successively greater energy. The ground state of the hydrogen atom, where its energy is lowest, is when the electron is in the orbit that is closest to the nucleus. When the electron is in one of these orbits, its energy is fixed. Carbon Bohr model The Bohr model of carbon contains a nucleus having 6 protons and 6 neutrons in the center, and around this nucleus, there are two electron shells containing 6 electrons. It accounts for a wide range of physical phenomena, including the existence of discrete packets of energy and matter, the uncertainty principle, and the exclusion principle.Īccording to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. It shows the atoms electrons in circular orbits at specific distances from the nucleus. Carbon Bohr model by Deep The information on this page is fact-checked. :max_bytes(150000):strip_icc()/Bohr-58e690203df78c51620ff02e.jpg)
This is a theory based on the principle that matter and energy have the properties of both particles and waves. This was the basis for what later became known as quantum theory. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy-a quantum of light, or photon. (Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window))īohr explained that electrons can be moved into different orbits with the addition of energy. Although these Bohr atomic models are sturdy, as with all polymer clay they need to be handled with care.\): Bohr's atomic model hydrogen emission spectra. Such variations are inherent in the manufacturing of handmade products, so you may expect minor distinctions that will make your Bohr Atomic Model Jewelry special and truly one of a kind.Ĭolor and contrast of the image may differ depending on your monitor settings. Due to this process, there may be a slight variation from one item to the next. Please note that our Bohr Atomic Model CO2 Carbon Dioxide 3D Jewelry Set are handmade by artisans one piece at a time. 
This allows the energy fields to spin independently from each other so the electrons can appear to orbit around the positively charged nucleus that is constructed with the appropriate number of Protons and Neutrons for each Element represented. 3d illustration RF ID: MC2N93 Preview Image details Contributor: Emilija Randjelovic / Alamy Stock Photo File size: 190.8 MB (1.2 MB Compressed download) Releases: Model - no Property - no Do I need a release Dimensions: 10000 x 6670 px 84.7 x 56.5 cm 33.3 x 22. .jpg)
Oxygen Earrings and Carbon Pendant are 2" in diameter and each "energy field" holding the electrons is free hanging which promotes movement. Carbon Atom Bohr model with proton, neutron and electron. 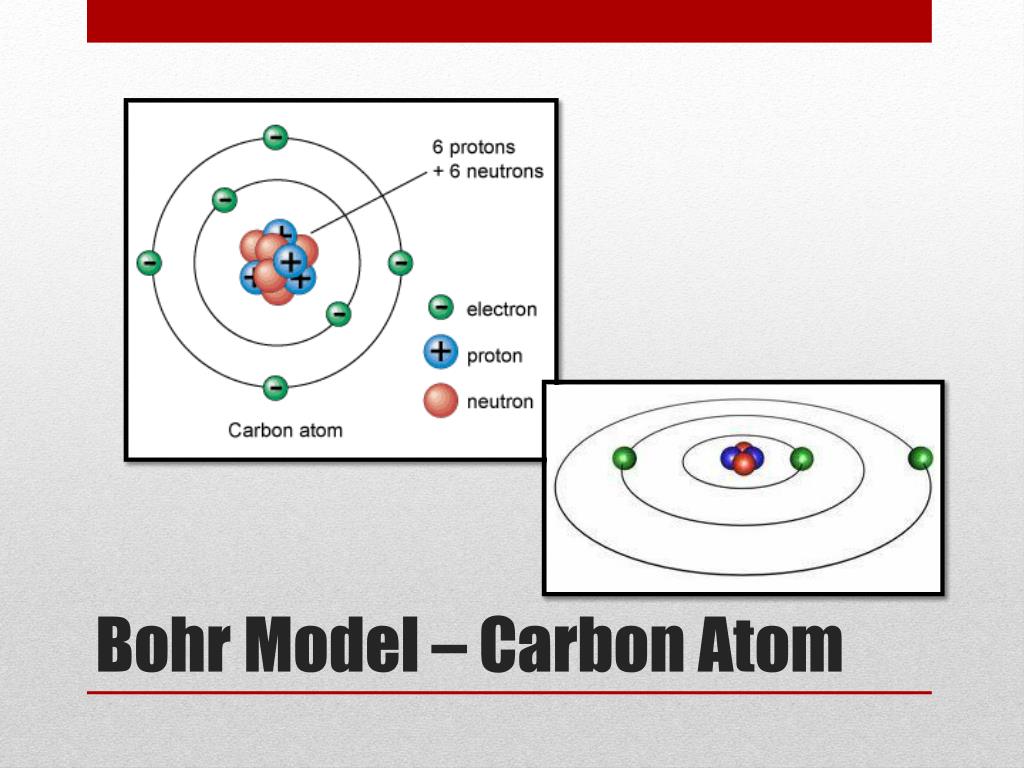
Pulled straight from the Periodic Table of Elements, these Bohr Atomic Model Oxygen and Carbon molecules make the perfect addition for any STEM fan, Science Teacher, or Periodic Table enthusiast's jewelry collection. Matter makes up everything so why not let it make your Jewelry? Show the universe your appreciation with this never Bohr-ing handmade representation of the Rutherford-Neil Bohr's Atomic Model that was introduced in 1913. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
